
Leave message
Can’t find what you’re looking for?
Fill out this form to inquire about our custom protein services!
Inquire about our Custom Services >>

































 Order Online! Now! Get your $50 coupon for online order.
Order Online! Now! Get your $50 coupon for online order. Order Online! Now! Get your $50 coupon for online order.
Order Online! Now! Get your $50 coupon for online order.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
 Fill out organ-on-a-chip questionnaire to win a FREE gift!
Fill out organ-on-a-chip questionnaire to win a FREE gift!  Fill out organ-on-a-chip questionnaire to win a FREE gift!
Fill out organ-on-a-chip questionnaire to win a FREE gift!
> Insights > SPR&BLI analytical service for the overall process of antibody drug development There is increasing use of biomolecular interaction analysis in drug development to kinetically characterize the binding activities of molecules. Based on the Biacore T200/8K platform and ForteBio Octet platform, ACROBiosystems provides SPR /BLI analytical services to our valued customers. The SPR/BLI analytical services include antibody screening, antibody pairing/group, kinetic analysis, and interaction measurement.
Relying on a strong and professional technical team, ACROBiosystems provides high-quality integrated SPR&BLI analytical services. ACROBiosystems has served and collaborated with more than 100 customers from academia and industry. Our work supported multiple antibody drugs IND applications. Here, ACROBiosystems Detection and Analysis Service Center will share how SPR&BLI technology can support you during the overall process for antibody drug development.
Case study
1. High-throughput antibody screening
You can do an initial affinity screening of your antibodies which helps to rank the candidates based on their specificities as well as kinetics. This can accelerate the therapeutic screening process.
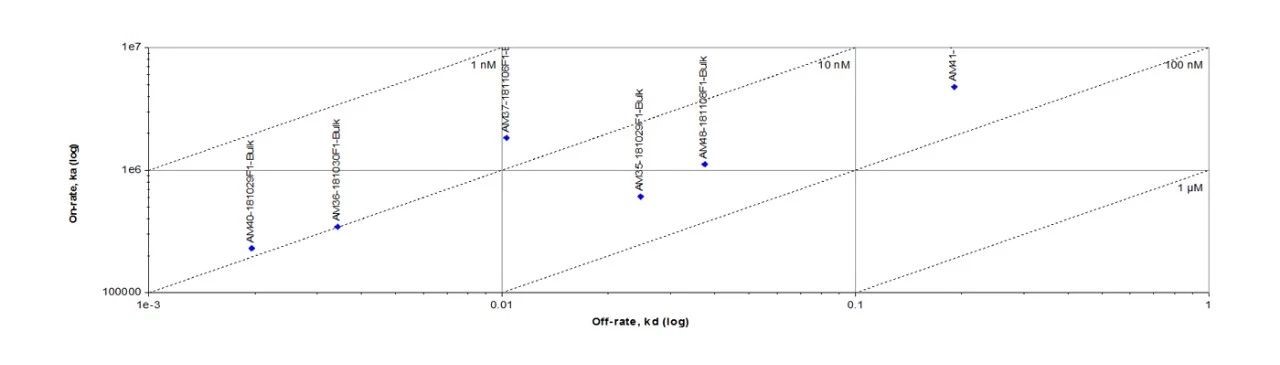
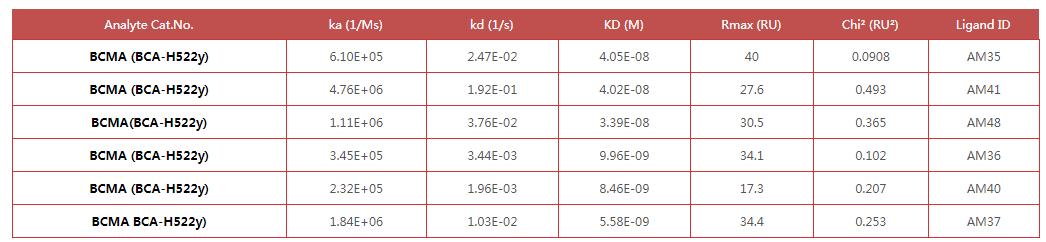
Fig. 1 Anti- BCMA antibodies (Mouse IgG1) captured on CM5 chip via anti-Mouse IgG antibody, can bind Human BCMA Protein, His Tag (Cat. No. BCA-H522y) as screened assay in a SPR (Biacore T200).
2. Antibody Characterization
Based on the multiple concentration affinity testing result, you can further understand the affinity and kinetic activities of the antibody, and predict the metabolism of the drug in vivo.
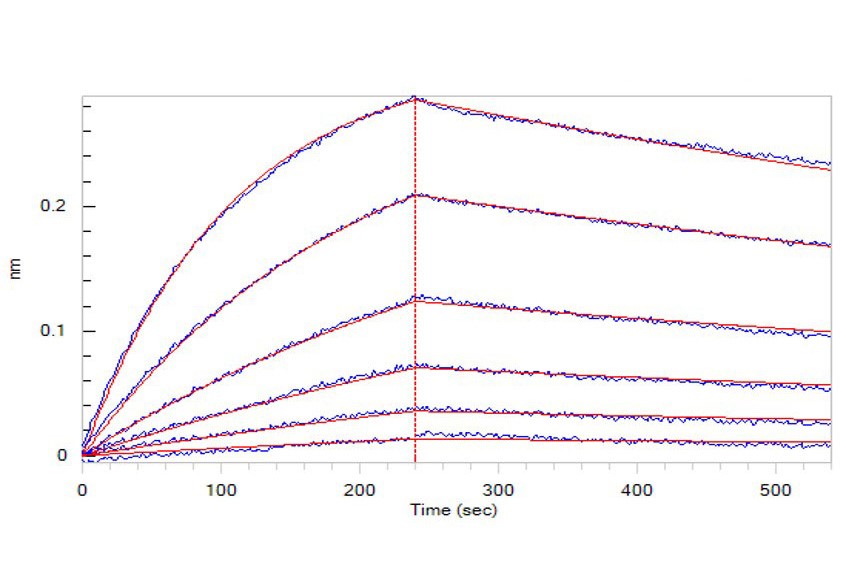
Fig.2 Opdivo (Nivolumab), loaded on Protein A biosensor, can bind to Human PD-1, His Tag (HPLC-verified) (Cat. No. PD1-H5221) with an affinity constant of 5.09 nM as determined in BLI assay (ForteBio Octet Red96e).
3. Batch-to-batch consistency evaluation
SPR technology can be used in quality control for batch-to-batch consistency during the therapeutic production process.
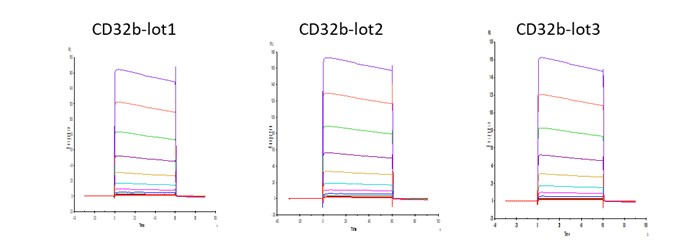
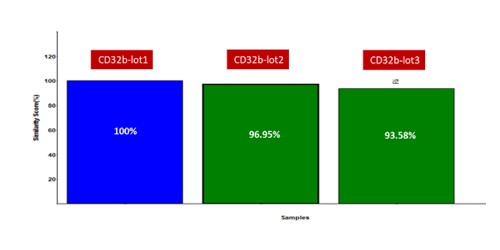
Fig.3 Batch-to-batch consistency result of Human Fc gamma RIIB / CD32b (Cat. No. CDB-H5228). CV is within 10% (Biacore T200).
4. Detection of affinity between the antibody and Fc receptor protein
The efficacy of therapeutic antibodies depends on not only the binding between the Fab fragments and target antigen, but also the binding between their Fc fragments and Fc receptors. Therefore, screening and optimization for antibodies with the most ideal affinity to Fc receptors are important to develop therapeutic monoclonal antibodies.
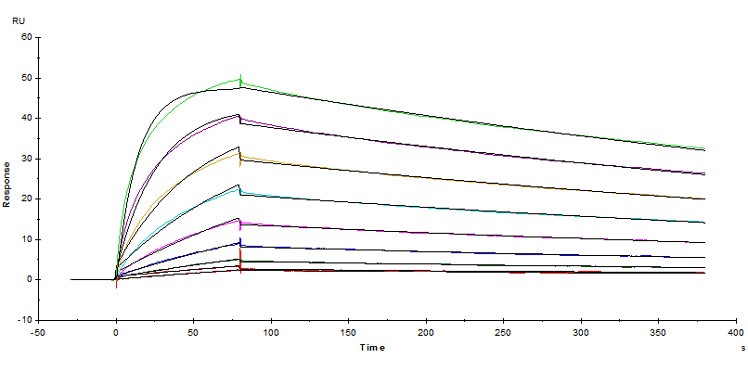
Fig.4 Immobilized Human Fc gamma RI / CD64 Protein (Cat. No. FCA-H52H2) on CM5 Chip via anti-Histidine antibody, can bind Herceptin® with an affinity constant of 5.45 nM as determined in SPR assay ( Biacore T200 ).
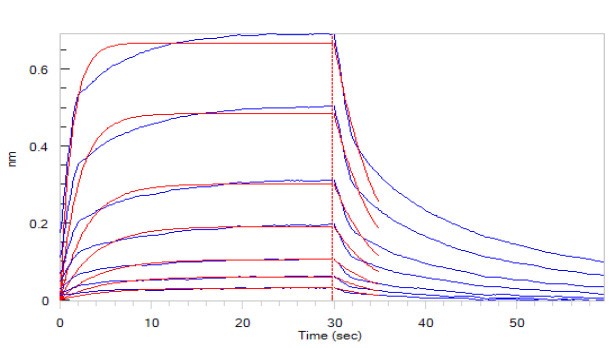
Fig.5 Human CD16a (F176), His Tag (SPR & BLI verified) (Cat. No. CDA-H5220) loaded on HIS1K Biosensor, can bind to Rituximab with an affinity constant of 1.72 μM as determined in BLI assay (ForteBio Octet Red96e) .
5. Cross-species reactivity screening
Antibodies may have different reactivities against antigens from various species. Cross-species reactivity screening can help to determine the most appropriate animal model for your animal studies.
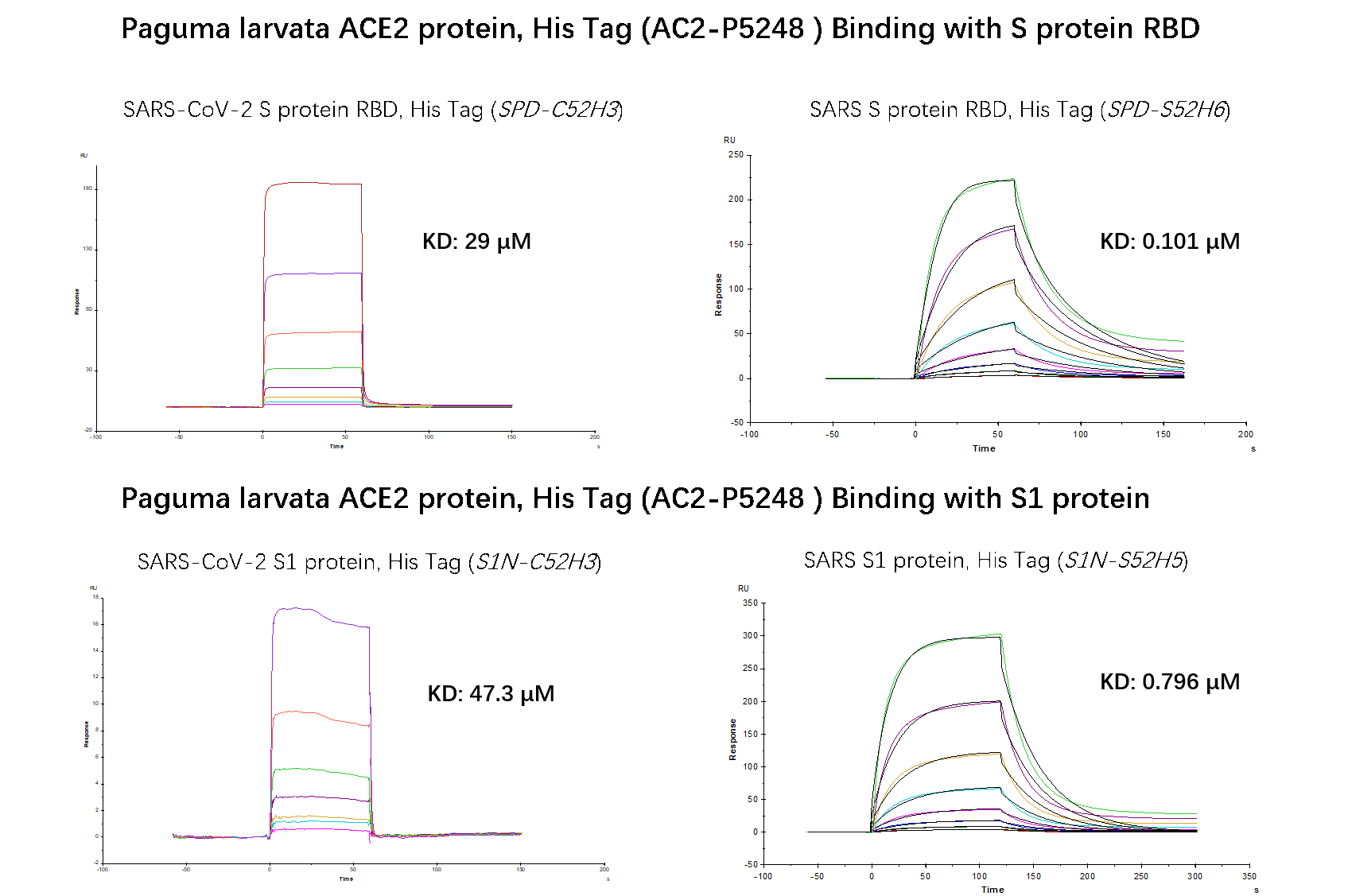
Fig.6 SPR verified that Paguma larvata ACE2 protein has a higher binding affinity to SARS than SARS-COV-2. The affinity constant of SARS S RBD and civet ACE2 can reach 0.101 µM, while the affinity constant of SARS-CoV-2 S RBD protein and civet ACE2 is only 29 µM. The affinity constant of SARS S1 protein and civet ACE2 can reach 0.796 µM, while the affinity constant of SARS-CoV-2 S1 protein and civet ACE2 is only 47.3 µM.
6. Bispecific antibodies
Bispecific antibody (BsAb) is a type of antibody molecule that can simultaneously bind to two different antigens or two different epitopes on the same antigen. Bispecific antibodies can exert the synergistic effect of two monoclonal antibodies, and may be more advantageous than mixed use of two monoclonal antibodies. The affinity measurement of the two Fab ends to their respective targets is also necessary for the molecule optimization.
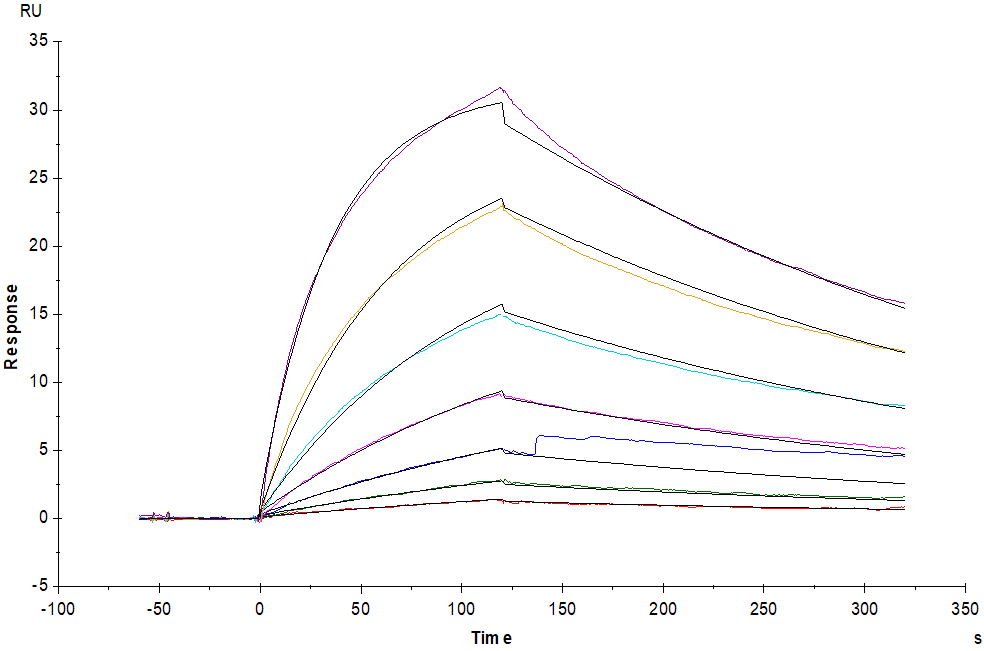
Fig.7 Bispecific T-cell Engager (CD3×BCMA) immobilized on CM5 Chip can bind to Human CD3E&CD3D Heterodimer Protein, His Tag&Tag Free (Cat. No. CDD-H52W1) with an affinity constant of 31.8 nM as determined in a SPR assay (Biacore T200).
7. Affinity measurement to complement systems
Most therapeutic antibodies kill or eliminate tumor cells through the CDC effect. The affinity between C1q protein complex and the antibody directly decides whether a stronge CDC effect can be induced. Therefore, affinity testing to C1q protein is also necessary in the development of antibody drugs.
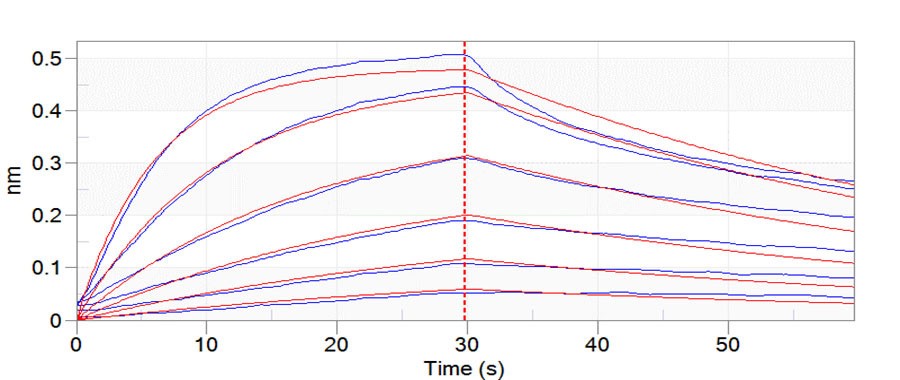
Fig.8 Trastuzumab can bind to C1q with an affinity constant of 3.24 nM as determined in BLI assay (ForteBio Octet Red96e) .
Promotion
All Fc receptor proteins needed are free of charge
Only provide analytes, then get the full service
Still have questions?
>ACROBiosystems’s experts are here to help.
>Call us at: +1 800-810-0816 or email at:
services@acrobiosystems.com
This web search service is supported by Google Inc.







