 Order Online! Now! Get your $50 coupon for online order.
Order Online! Now! Get your $50 coupon for online order. Order Online! Now! Get your $50 coupon for online order.
Order Online! Now! Get your $50 coupon for online order.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
 Fill out organ-on-a-chip questionnaire to win a FREE gift!
Fill out organ-on-a-chip questionnaire to win a FREE gift!  Fill out organ-on-a-chip questionnaire to win a FREE gift!
Fill out organ-on-a-chip questionnaire to win a FREE gift!
> Insights > Classical, Potential, and Innovative Therapeutic Strategies for Brain Tumors Brain tumors account for approximately 1.35% of all malignancies and 29.5% of cancer-related deaths. According to their origin, brain tumors can be classified as primary or metastatic. Approximately 70% of brain tumors are benign and others are malignant. The most common malignant brain tumor is glioblastoma, which originates from glial cells. The five-year survival rate of glioblastoma is less than 5%, with an average survival rate of one year. The median age of diagnosis for a primary brain tumor is 60 years old, with an average survival rate of 75.2%. For patients with benign brain tumors, the average five-year survival rate is 91.7%, while the relative five-year survival rate for patients with malignant brain tumors is 36% after diagnosis. Moreover, it is estimated that one-third of patients with primary malignancies develop metastatic CNS lesions during the course of their disease.
According to Neurology, a sub-journal of Lancet, China ranks first in the world in terms of incidence and mortality of brain tumors, with an annual incidence rate of 7/100,000, accounting for 2% of all tumors. Furthermore, as the brain is caged by the skull, there is little space for the tumor to grow. Therefore, the tumor will compress the surrounding nerves, blood vessels, and tissues. Consequently, it affects the brain functions, causing weakness, difficulty in walking, loss of balance, partial or complete blindness, and difficulty in understanding or using language, memory problems, etc. As compared to other types of tumors, brain tumors, whether malignant or not, often have a more lasting and severe impact on the patients’ life quality.
The common treatments for brain tumors include chemotherapy, radiation therapy, and surgery. Meanwhile, immunotherapy, which stimulates patients’ own immune system to eliminate tumor cells, has been widely studied as a potential treatment option. Currently, the development of new immunotherapeutic drugs for brain tumors, especially glioblastoma, focuses on various types of drugs including small molecule drugs, vaccines, monoclonal antibodies, bispecific antibodies, fusion proteins, ADC, and CAR-T targeting EGFR, VEGF, c-kit(CD117), TOP, PDGF, CSF-1, FGF, mTOR, PI3K, and Fit3.
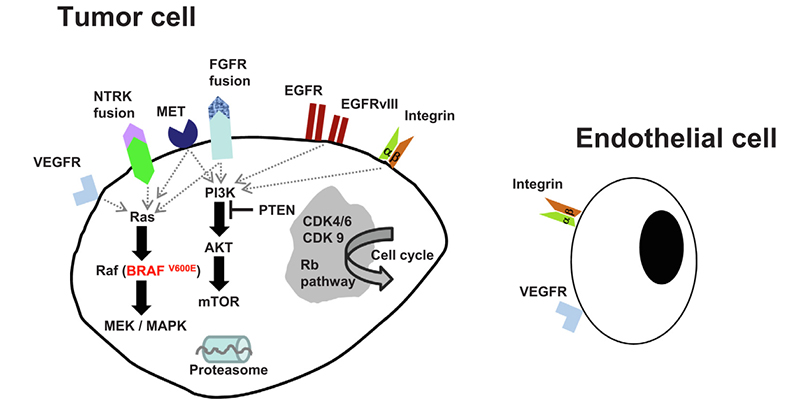
Candidate molecular pathways targeting interventions in glioblastoma
For example, EphA2 is abundantly expressed in glioblastoma and low in normal brain tissue, while CAR - T cell therapy targeting EphA2 is an attractive strategy to improve the outcomes of therapy. We introduce the classic and potential therapeutic strategies for brain tumors as well as the innovative therapy.
1. Classical Therapeutic Strategy: Inhibition of Brain Tumor Growth
The function of tyrosine kinase is closely related to tumorigenesis and development. More than 50% of proto-oncogenes and oncogene products are tyrosine kinases, and their abnormal expression usually leads to disruption of cell proliferation regulation, resulting in tumorigenesis. In addition, abnormal tyrosine expression is also associated with tumor invasion, metastasis, tumor neovascularization, and tumor chemoresistance. More than 20 different families of receptor and non-receptor tyrosine kinases are currently being used as targets for antitumor drug development, including EGFR, VEGFR, PDGFR, FGFR, etc.
Taking Nimotuzumab as an example, the anti-EGFR inhibitor is widely used in brain tumors. However, it has slightly improved overall survival when used in children with high-grade gliomas.
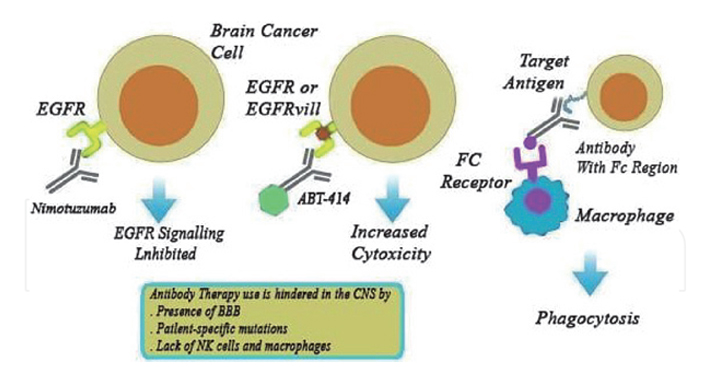
Mechanism of anti-EGFR monoclonal antibody in the treatment of brain tumors
The VEGF/VEGFR signaling promotes neovascularization and enhances tumor growth. Therefore, targeting this signaling pathway is an important direction to inhibit tumor neovascularization in antitumor-targeted drug research. Notably, the monoclonal antibody targeting VEGF, bevacizumab, has been approved for the indication of glioblastoma in many regions worldwide.
To some extent, monoclonal antibody treatment in the brain has suboptimal survival benefits because monoclonal antibodies are unable to penetrate the blood-brain barrier (BBB). So, one of the limits is that they can’t cause considerable barrier disruption. Furthermore, patient-specific antigen mutations affect antibody binding efficiency.
2. Potential Therapeutic Strategy: Targeting Intracellular Signaling
The PI3K-AKT-mTOR pathway, consisting of phosphatidylinositol 3-kinase (PI3K), its downstream protein kinase B (PKB/Akt) and target of rapamycin protein (mTOR) is an intracellular signaling pathway that plays a pivotal role in the cell cycle control. The Ras-MAPK pathway, consisting of the serine protein kinase Ras and the mitogen-activated protein kinase (MAPK) tertiary cascade kinase, is important in transducing signals from the extracellular milieu to the cell nucleus. These pathways, their downstream signal transduction and transcriptional activation together with the STAT family of factors are closely related to tumorigenesis and progression.
In glioblastoma, the positive expression rate of PI3K/AKT/mTOR is highly associated with the clinical grade and the prognosis of the disease. Therefore, the PI3K-AKT-mTOR signaling pathway has emerged as a promising therapeutic strategy against brain tumors.
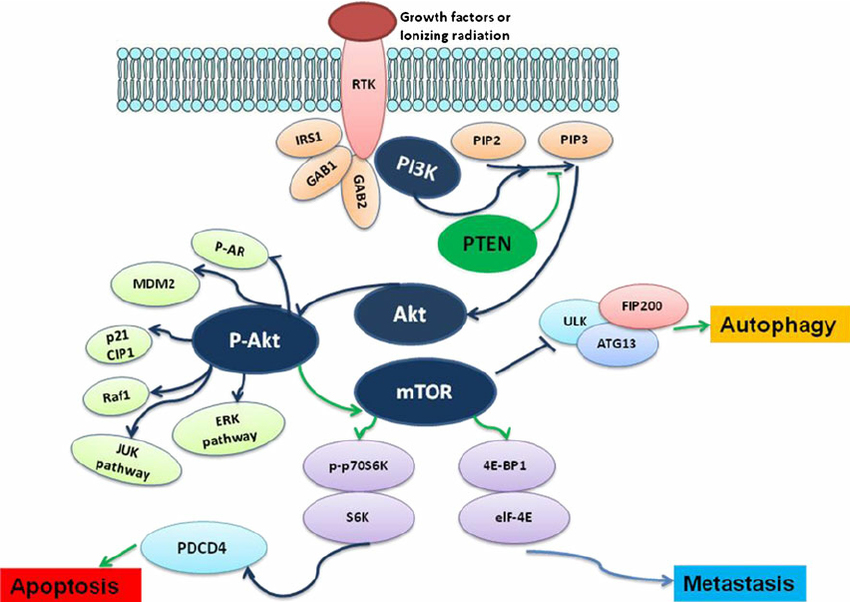
The role of PI3K-AKT-mTOR signaling pathway in the regulation of tumor metastasis, apoptosis, and autophagy
A number of small-molecule inhibitors targeting the PI3K-AKT-mTOR signaling pathway have entered phase I clinical studies. These inhibitors include wortmannin, LY294002, IC484068, which target the catalytic subunit p110 of PI3K; the natural origin PI3K inhibitor fisetin (deguelin); straurosporine, UCN-01, and perifosine, inhibitors of PDK, a serine/threonine kinase necessary for Akt activation; and rapamycin and its analogs RAD001, CI779 and AP23573, inhibitors that specifically target mTOR. Among them, rapamycin and its derivatives that specifically target downstream mTOR molecules have demonstrated better therapeutic effects in clinical trials and have shown good development prospects.
3. Innovative Therapeutic Strategy: Anti-brain Tumor Vaccines
Due to the presence of special proteins on the surface of brain tumor cells, vaccines targeting these proteins can enable the immune system to specifically eliminate tumor cells without harming normal cells.
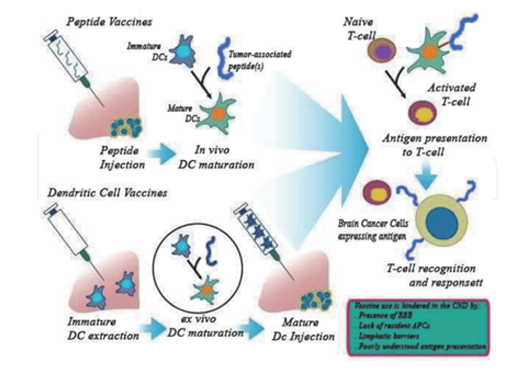
TPolypeptide vaccines and dendritic virus vaccines for the treatment of brain tumors
SurVaxM, a world-first peptide-mimetic tumor vaccine has demonstrated safety and tolerability in a phase I study in patients with malignant glioma. It is made from natural amino acids that break down when an immune response is generated resulting in minimal side effects. The latest results from the phase II study show that SurVaxM vaccine has a safety profile, good tolerability, and the ability to prolong survival even in the most difficult-to-treat patient subgroup. In 2017, SurVaxM vaccine was granted orphan drug by the FDA.
AV-GBM-1 is an autologous specific dendritic cell vaccine. The vaccine carries specific antigenic information extracted from post-operative tumor tissue, once injected, it will deliver the antigenic information to T cells to stimulate tumor-killing activity. A phase II clinical data for AV-GBM-1 were announced on April 8, 2020, showing that this novel vaccine has great potential to extend overall survival in patients with newly diagnosed glioblastoma. The phase II clinical trial data presented at the ASCO meeting in June 2021 showed that this vaccine has the potential to extend overall survival in patients with newly diagnosed glioblastoma and significantly improved progression-free survival over standard treatment regimens.
IGV-001 is an autologous tumor cell vaccine that uses the patient's immune system as a defense mechanism to target tumor cells in multiple manners. Firstly, it uses the surgically removed tumor cells from glioblastoma patients. These cells are further treated with IGF-1R antisense oligonucleotides (IMV-001) to push the tumor cells toward controlled cell death. Subsequently, these treated cells are again mixed with an excess of IMV-001 (as adjuvant) in an infiltration chamber to be finally implanted into the patient's abdomen. These cells have to be implanted within 24 hours of surgery and to be removed after 48 hours of implantation. This innovative vaccine has been granted orphan drug by the FDA as well.
ACRO is developing more proteins for the diagnosis and treatment. If you have any needs or more development suggestions, don't hesitate to contact us.
1. Le Rhune, et al. Molecular targeted therapy of glioblastoma. Cancer Treat Rev, 2019. https://doi.org/10.1016/j.ctrv.2019.101896.
2. Manimekalai Pichaivel, Gayathri Anbumani, Panneerselvam Theivendren and Muruganantham Gopal. An Overview of Brain Tumor. 2022. DOI:10.5772/intechopen.100806
3. Chang, Lei & Graham, Peter & Hao, Jingli & Bucci, Joseph & Cozzi, Paul & Kearsley, John & li, Yong. Emerging roles of radioresistance in prostate cancer metastasis and radiation therapy. Cancer metastasis reviews. https://doi.org/10.1007/s10555-014-9493-5
4. Platten, M., Bunse, L., Wick, A. et al. A vaccine targeting mutant IDH1 in newly diagnosed glioma. Nature 592, 463–468 (2021). https://doi.org/10.1038/s41586-021-03363
5. https://medicalxpress.com/news/2021-03-first-ever-vaccine-malignant-brain-tumors.html
This web search service is supported by Google Inc.
