 Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit! Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit!
 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
| Cat. No. | Species | Product Description | Structure | Purity | Feature |
|---|---|---|---|---|---|
| ILA-H82F5 | Human | Biotinylated Human IL-5 R alpha / CD125 Protein, Avitag™,Fc Tag (MALS verified) |  |
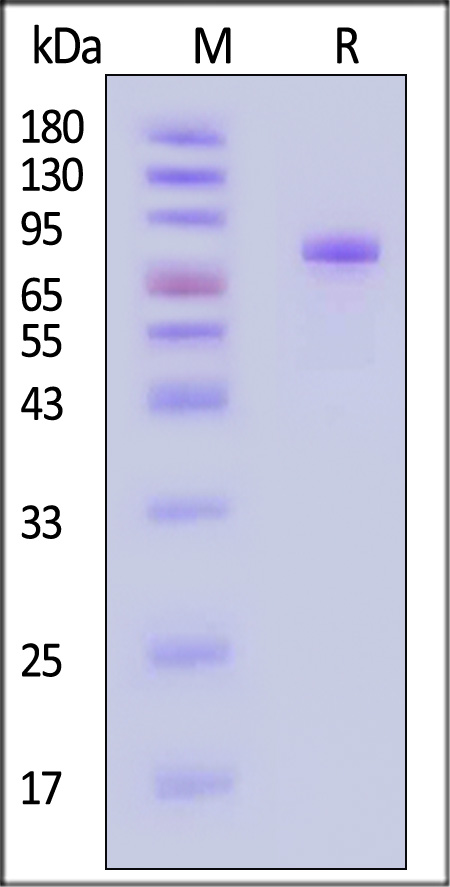
|

|
| ILA-H5269 | Human | Human IL-5 R alpha / CD125 Protein, Fc Tag (MALS verified) |  |

|

|
| ILA-H52H4 | Human | Human IL-5 R alpha / CD125 Protein, His Tag (MALS verified) |  |

|

|

Loaded Biotinylated Human IL-5, His,Avitag (Cat. No. IL5-H82Q5) on SA Biosensor, can bind Human IL-5 R alpha, His Tag (Cat. No. ILA-H52H4) with an affinity constant of 12.6 nM as determined in BLI assay (ForteBio Octet Red96e) (QC tested).
| Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| Benralizumab | BIW-8405; KHK-4563; MEDI-563; BIW-8405-IL-5R | Approved | Biowa Inc, Medimmune Llc | Fasenra | United States | Asthma | Astrazeneca Ab | 2017-11-14 | Genetic Diseases, Inborn; Gastroenteritis; Skin Diseases, Genetic; Hypersensitivity; Eczema; Dermatitis, Atopic; Hypersensitivity, Immediate; Pulmonary Disease, Chronic Obstructive; Leukocyte Disorders; Sinusitis; Pemphigoid, Bullous; Asthma; Eosinophilic Esophagitis; Bronchiectasis; Eosinophilic gastroenteritis (EG); Skin Diseases, Eczematous; Dermatitis; Skin Diseases; Immune System Diseases; Chronic Urticaria; Respiratory Tract Diseases; Eosinophilia; Nasal Polyps; Asthma, Exercise-Induced; Eosinophilic Granulomatosis With Polyangiitis; Hematologic Diseases; Hypereosinophilic Syndrome | Details |
| Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| CSL-311 | CSL-311 | Phase 1 Clinical | Csl Ltd | Asthma | Details |
This web search service is supported by Google Inc.
