 Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit! Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit!
 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!

Imagine the capability of culturing a complex collection of cells that mimics a patient’s cellular tissue environment and by extension, organs. In recent years, this has become a reality through ‘organoids’, three-dimensional constructs, that imitate the architecture and functionality of native organs. Organoids contain self-renewing stem cell populations, which can differentiate into specific cell types in organ tissues. Compared with traditional 2D cell culture, organoids have richer cell composition and physiological function closer, therefore finding widespread use in biological function research, artificial organ development, disease modeling and drug screening.
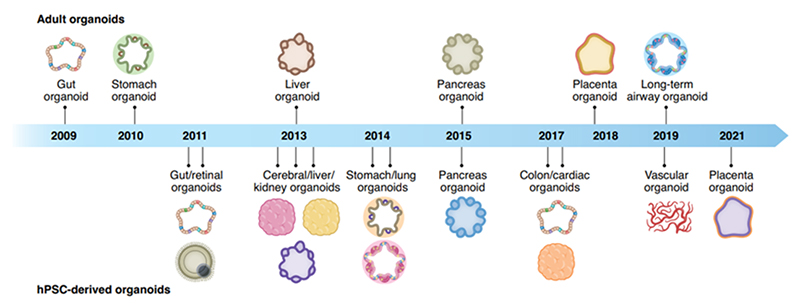
Figure 1. Organoid discovery of their corresponding organs through the years. 1
There are two main sources of organoids: adult stem cells (ASCs) and pluripotent stem cells (PSCs), whereas the latter includes embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs). ASCs refer to cell types that maintain stem cell potential in adult human organs, which can promote the homeostatic maintenance or damage repair process of body organs. Overall, ASCs are organ-specific, and its organoids are accurate representations of both structural and functional characteristics of adult tissues. PSCs, on the other hand, need to undergo multiple differentiation processes. This simulates the entire process of organ development, making it a critical tool in organ development and gene function studies. Currently, both stem cell sources can form most of the major organs. However, ASCs are more widely used to construct organs and tissues. Several exceptions are for tissue samples that are harder to obtain such as brain tissue. In these cases, PSCs are significantly more advantageous for organoid model building. Despite the differences between the stem cell sources, growth factors play a critical role in inducing and directing cell differentiation. Organoid culturing typically contains multiple growth factors and can be highly variable between organoid types. As such, organoids research requires an optimized extracellular matrix composition ensure proper physiological development.
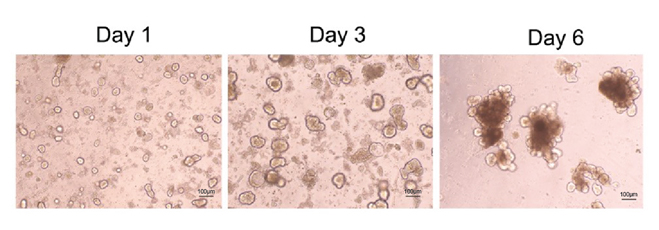
Figure 2. Confocal microscopy of mouse intestinal organoid growth with growth factors: human EGF (Cat. EGF-H52H3), Noggin (Cat. NON-H5257), and R-spondin1 (Cat. RS6-H4220).
Currently, organoids play an important role in three areas of research: precision medicine, drug discovery and regenerative medicine. In precision medicine, organoid models are optimal for high-throughput drug screening. A biopsy is taken from a patient for organoid culturing, where the cultured organoid is then exposed to various xenobiotics to screen for the optimal therapeutic effect, enabling personalized medicine. Organoids have been proven to be excellent drug screening models in a variety of tumor types, such as colorectal cancer, breast cancer, non-small cell lung cancer, gastric cancer, etc. According to several research articles, the accuracy of organoid drug response prediction in a variety of tumors can reach 80%, showing immense potential in the implementation of personalized medicine for diagnostic use.
In drug development, organoids represent a modality that is more accurate than conventional cell lines and more cost-effective than animal models. Its complex cell composition, tissue structure, and physiological structure along with the high repeatability and throughput of in vitro studies make it an ideal model for preclinical drug toxicity evaluation. At the same time, gene editing can be used to build a variety of disease models, such as cystic fibrosis, colorectal cancer, microcephaly, and other diseases. In addition, organoids can also be used to study host-microbial infection processes, such as the invasion of COVID-19 and Heliobacter pylori. In recent years, due to the COVID-19 pandemic, scientists have constructed a variety of organ models to study the infection process and the physiological function changes brought by infection. Similarly, the world's top 20 pharmaceutical companies have all participated in the research and development of organoid applications, including Novartis, Pfizer, and Johnson & Johnson, to test the safety and effectiveness of their novel drugs.
In regenerative medicine, organoids, as larger, more mature stem-cell models, have also been studied to explore the potential to repair damaged tissues and organs. As such, several studies have explored the transplantation of organoids to combat inflammatory bowel disease and treat short bowel syndrome. Several studies have also been conducted to attenuate type I diabetes by constructing islet organoids, but are currently limited to preclinical animal models. However, these studies are still in the early stage. Key problems such as heterogeneity of cultured organoids, the potential tumorigenicity of using matrix gel in organoid culture, and the homing effect in vivo after organoid transplantation are all concerns for clinical use. Despite these issues, the potential for organoids in regenerative medicine makes it a focal point in organoids research.
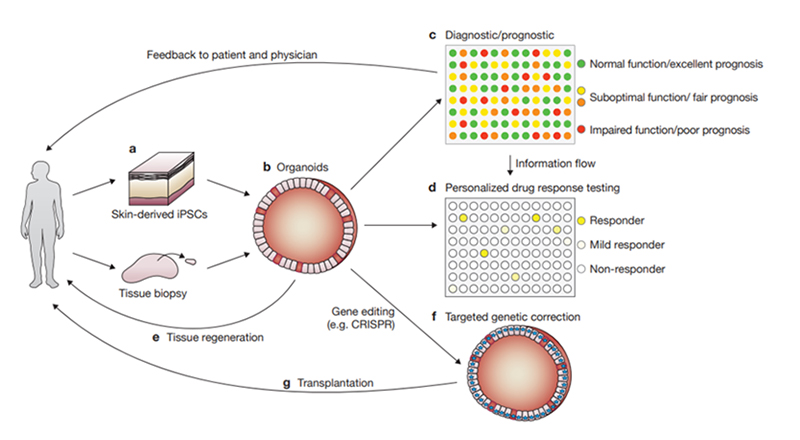
Figure 3. Various applications of organoids for disease research, drug development, and personalized medicine. 2
Organoids represent the next step beyond two-dimensional cell culturing, simulating the architectural and functional characteristics of human organs and even the tumor microenvironment. With recent advancements in organoid culturing and elucidation of growth factors needed to direct cell differentiation, organoids have become an indispensable tool in the process of drug development and evaluation. Currently, most organs in the human body can be formed into organoids in vitro, reproducing the liver, lung, heart, blood vessels, and many others. As such, organoids have the potential to become an essential preclinical model for human disease research and drug development.
To support research on organoid cell culturing, ACROBiosystems has developed a series of high-quality ActiveMax® cytokines, including EGF, Noggin, R-Spondin 1, FGF10, FGF2, and Activin A. These products are suitable as growth factors for organoid culture that have been verified to promote organoid growth.
References:
1. Han Y., Chen S., et al. Human Organoid Models to Sutdy SARS-CoV-2 Infection. Nature Methods. 2022, 19,418-428.
2. Fatehullah A., Barker N., et al. Organoids as an In Vitro Model of Human Development and Disease. Nat Cell Biol. 2016, 18(3):246-254.
This web search service is supported by Google Inc.
