 Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit! Request a FREE Sample of our Fc gamma RI / CD64 Binding Kit !
Request a FREE Sample of our Fc gamma RI / CD64 Binding Kit !
 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
| Cat. No. | Species | Product Description | Structure | Purity | Feature |
|---|---|---|---|---|---|
| TTR-H5223 | Human | Human Transthyretin / Prealbumin Protein, His Tag |  |
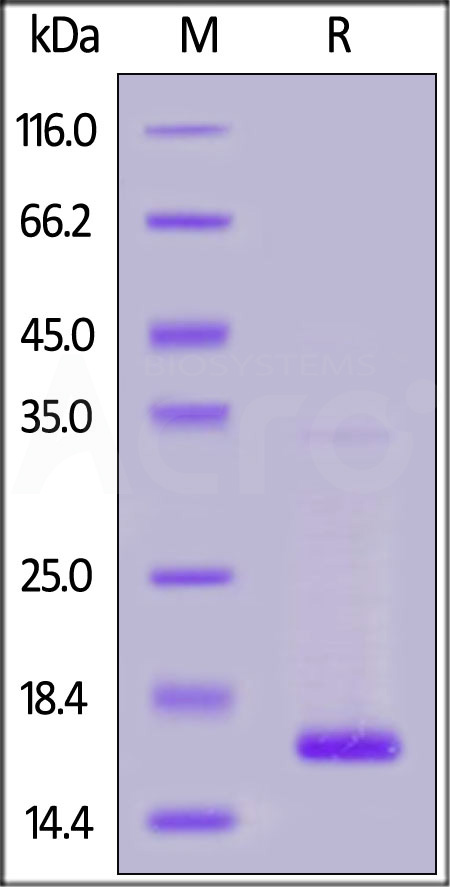
|
| Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| Patisiran | SAR-438037; GENZ-438027; ALN-TTR02; ALN-18328 | Approved | Alnylam Pharmaceuticals Inc | Onpattro | United States | Amyloid Neuropathies, Familial | Alnylam Pharmaceuticals Inc | 2018-08-10 | Amyloidosis, Familial; Amyloid Neuropathies, Familial; Polyneuropathies; Cardiomyopathies; Amyloidosis; Transthyretin amyloidosis; Amyloid Neuropathies | Details |
| Vutrisiran | ALN-TTRSC02 | Approved | Alnylam Pharmaceuticals Inc | AMVUTTRA | United States | Amyloid Neuropathies, Familial; Transthyretin amyloidosis | Alnylam Pharmaceuticals Inc | 2022-06-13 | Amyloidosis, Familial; Amyloid Neuropathies, Familial; Cardiomyopathies; Amyloidosis; Transthyretin amyloidosis | Details |
| Inotersen sodium | ISIS-420915; GSK-2998728 | Approved | Ionis Pharmaceuticals Inc | Tegsedi | EU | Amyloidosis | Akcea Therapeutics Ireland Ltd | 2018-07-05 | Amyloid Neuropathies, Familial; Adenomatous Polyposis Coli; Amyloidosis; Transthyretin amyloidosis | Details |
| Tafamidis | PF-6291826; Fx-1006A; PF-06291826; FX-1006A | Approved | Pfizer Inc | 维万心, VYNDAMAX | United States | Heart Diseases | Foldrx Pharmaceuticals Inc | 2019-05-03 | Heart Diseases; Transthyretin amyloidosis | Details |
| Eplontersen | ION-682884; ION-TTR-LRx; IONIS-TTR-LRx; AKCEA-TTR-LRx | Approved | Ionis Pharmaceuticals Inc | WAINUA | United States | Transthyretin amyloidosis | Astrazeneca Ab | 2023-12-21 | Amyloid Neuropathies, Familial; Cardiomyopathies; Transthyretin amyloidosis | Details |
| Tafamidis Meglumine | FX-1006A; PF-6291826; Fx-1006A; PF-06291826 | Approved | Pfizer Inc | 维达全, Vyndaqel | EU | Amyloidosis | Pfizer Europe Ma Eeig | 2011-11-16 | Amyloid Neuropathies, Familial; Immunoglobulin Light-chain Amyloidosis; Cardiomyopathies; Amyloidosis; Transthyretin amyloidosis; Amyloid Neuropathies | Details |
| Patisiran | SAR-438037; GENZ-438027; ALN-TTR02; ALN-18328 | Approved | Alnylam Pharmaceuticals Inc | Onpattro | United States | Amyloid Neuropathies, Familial | Alnylam Pharmaceuticals Inc | 2018-08-10 | Amyloidosis, Familial; Amyloid Neuropathies, Familial; Polyneuropathies; Cardiomyopathies; Amyloidosis; Transthyretin amyloidosis; Amyloid Neuropathies | Details |
| Vutrisiran | ALN-TTRSC02 | Approved | Alnylam Pharmaceuticals Inc | AMVUTTRA | United States | Amyloid Neuropathies, Familial; Transthyretin amyloidosis | Alnylam Pharmaceuticals Inc | 2022-06-13 | Amyloidosis, Familial; Amyloid Neuropathies, Familial; Cardiomyopathies; Amyloidosis; Transthyretin amyloidosis | Details |
| Inotersen sodium | ISIS-420915; GSK-2998728 | Approved | Ionis Pharmaceuticals Inc | Tegsedi | EU | Amyloidosis | Akcea Therapeutics Ireland Ltd | 2018-07-05 | Amyloid Neuropathies, Familial; Adenomatous Polyposis Coli; Amyloidosis; Transthyretin amyloidosis | Details |
| Tafamidis | PF-6291826; Fx-1006A; PF-06291826; FX-1006A | Approved | Pfizer Inc | 维万心, VYNDAMAX | United States | Heart Diseases | Foldrx Pharmaceuticals Inc | 2019-05-03 | Heart Diseases; Transthyretin amyloidosis | Details |
| Eplontersen | ION-682884; ION-TTR-LRx; IONIS-TTR-LRx; AKCEA-TTR-LRx | Approved | Ionis Pharmaceuticals Inc | WAINUA | United States | Transthyretin amyloidosis | Astrazeneca Ab | 2023-12-21 | Amyloid Neuropathies, Familial; Cardiomyopathies; Transthyretin amyloidosis | Details |
| Tafamidis Meglumine | FX-1006A; PF-6291826; Fx-1006A; PF-06291826 | Approved | Pfizer Inc | 维达全, Vyndaqel | EU | Amyloidosis | Pfizer Europe Ma Eeig | 2011-11-16 | Amyloid Neuropathies, Familial; Immunoglobulin Light-chain Amyloidosis; Cardiomyopathies; Amyloidosis; Transthyretin amyloidosis; Amyloid Neuropathies | Details |
| Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| NTLA-2001 | NTLA-2001 | Phase 3 Clinical | Intellia Therapeutics Inc | Amyloidosis, Familial; Cardiomyopathies; Amyloid Neuropathies; Transthyretin amyloidosis | Details |
| NNC6019-0001 | NNC6019-0001; NNC-6019; PRX-004 | Phase 2 Clinical | Novo Nordisk A/S | Cardiomyopathies; Transthyretin amyloidosis | Details |
| NI-006 | NI-006 | Phase 1 Clinical | Neurimmune Ag | Cardiomyopathies; Transthyretin amyloidosis | Details |
| NTLA-2001 | NTLA-2001 | Phase 3 Clinical | Intellia Therapeutics Inc | Amyloidosis, Familial; Cardiomyopathies; Amyloid Neuropathies; Transthyretin amyloidosis | Details |
| NNC6019-0001 | NNC6019-0001; NNC-6019; PRX-004 | Phase 2 Clinical | Novo Nordisk A/S | Cardiomyopathies; Transthyretin amyloidosis | Details |
| NI-006 | NI-006 | Phase 1 Clinical | Neurimmune Ag | Cardiomyopathies; Transthyretin amyloidosis | Details |
This web search service is supported by Google Inc.
