Prostate-specific membrane antigen (PSMA), a transmembrane glycoprotein located on the cell membrane, is specifically and highly expressed in prostate cancer (PCa). Besides, its expression level is related to tumor invasiveness. As a molecular target of PCa, PSMA has been extensively studied in the past two decades. Prostate cancer is one of the most common malignant tumors in Europe and the United States. Not only PSMA is highly expressed in advanced and castration-resistant prostate cancers, but the large extracellular domain makes it a perfect target for immune methods. In addition, the results of PSMA-targeted radionuclide therapy also confirm that it is an ideal target for the diagnosis and treatment of prostate cancer.
For the treatment of advanced prostate cancer, androgen deprivation therapy (ADT) is still the first-line therapy, but advanced prostate cancer often develops metastatic or castration resistance, which in turn develops into Castrate-Resistant Prostate Cancer (CRPC). For CRPC, androgen receptor blockers (AR) such as abiraterone and enzalutamide have been widely used in clinical practice, but the remission rate is limited. Therefore, more therapies for PSMA need to be explored. Currently, a great deal of evidence suggests that significant progress has been made in the PSMA-targeted therapy of PCa. It is expected to provide new drug options for patients with advanced castration-resistant prostate cancer in the future.
Targeted PSMA immunotherapy is mainly divided into the following categories: Antibody-drug conjugates (ADCs), chimeric antigen receptor T cells (CAR-T) and bispecific T cell redirection therapy.
The selection of antigens is a key factor in the drug development process for ADCs. It is necessary to satisfy the characteristics of not expressing or low expression in healthy cells but expressing on the surface of tumor cells that ultimately leads to promoting the internalization of ADC transport into cells. PSMA meets these requirements and ADCs targeting PSMA have made good progress so far.
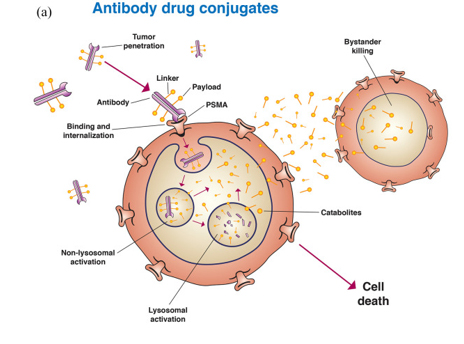
Image source: PSMA targeting in metastatic castration-resistant prostate cancer: where are we and where are we going? Doi: 10.1177/17588359211053898
On August 3, 2021, Ambrx Announced that antibody-conjugated drug (ADC), ARX517, which targets PSMA, has completed its first patient dosed in Phase 1 clinical trial. The multicenter, open-label, dose-escalation Phase 1 clinical trial enrolled 76 patients with advanced solid tumors who have failed previous standard therapy. They had assessed the safety, pharmacokinetics (PK), and antineoplastic activity of ARX517 in patients with tumors expressing PSMA in the treatment of prostate, pancreatic, lung, and ovarian cancers. In addition, on September 30, Hengrui Pharmaceutical also published a new drug patent for ADCs targeting PSMA.
However, there are also results that the occurrence of treatment-related adverse events may limit the therapeutic capacity of ADCs targeting PSMA in CRPC. MlN2704, for example, is an ADC that conjugates a humanized J591 antibody to Maytansine analog drug maytansinoid-1 (DM1). In a study of 62 patients treated with different regimens, 5 patients had a PSA50 response and adverse events, including peripheral neuropathy, were observed.
>>Click here to explore more ADC target proteins and download the free protocols
Another emerging field that has shown promise is chimeric antigen receptor T-cell (CAR-T) therapy. CAR-T therapy involves taking a patient’s T cells, engineering them to express a T cell receptor directed against a certain antigen, expanding the cells, and reinfusing them back into the patient. On August 31, Poseida Therapeutics announced that its autologous CAR-T cell therapy, P-PSMA-101, has yielded positive preliminary results in Phase 1 clinical trial for the treatment of patients with metastatic castration-resistant prostate cancer (mCRPC). In the trial, the therapy showed encouraging activity in the first 9 patients who received low-dose treatment. P-PSMA-101 is a CAR-T cell candidate that targets PSMA by genetically engineering the patient's own T cells so that they can safely and effectively eliminate tumor cells.
The results of a Phase I trial of CAR-T therapy targeting PSMA showed that five patients received PSMA-targeted CAR-T cells and sustained infusions of low-dose IL-2 after pretreatment chemotherapy. Two of the patients achieved the PSA50 response, no patients were observed with CRS, and the use of PSMA-specific CAR-T cells plus IL-2 resulted in a more pronounced antitumor response than monotherapy.
Another trial of 7 patients with prostate cancer showed that patients received PSMA-targeted CAR-T therapy after pretreatment chemotherapy. CAR-T cells persisted in the blood for up to 2 weeks, and one patient was stable for > 16 months.
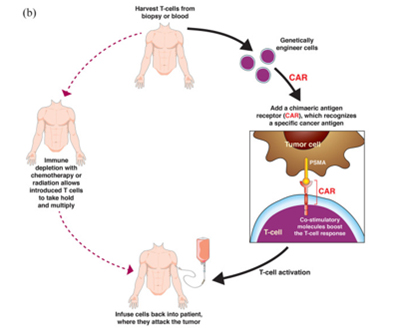
Image Source: PSMA targeting in metastatic castration-resistant prostate cancer: where are we and where are we going? Doi: 10.1177/17588359211053898
Although CAR-T therapy is surprisingly effective in blood tumors, however, response rates are lower in solid tumors. While PSMA appears to be an ideal target for CAR-T cell therapy, there are still potential barriers such as immunosuppressive TME to overcome.
>>Click here to explore more CAR-T target proteins and download the free protocols
Bispecific T-cell engagers (BiTEs) are another evolving area in prostate cancer treatment. BiTEs are bispecific monoclonal antibodies, one of which binds to T-cells through the CD3 receptor, the other of which is targeted against a tumor-specific marker. AMG 212 is a dual antibody that targets CD3 and PSMA, and one study enrolled 16 patients with mCRPC, divided into 5 different dose groups. At the time of the data report, PSA levels in 3 patients decreased by more than half. In one of the patients who had been treated for 19.4 months, there was a complete regression of the soft tissue metastases and a marked regression of bone metastases. However, as with other bispecific drugs that targeted CD3, AMG 212 had a short half-life and required continuous intravenous infusion. Amgen and BeiGene jointly declared a new drug, AMG160, for the treatment of metastatic castration-resistant prostate cancer (mCRPC) in adults. AMG 160 is new HLE-BiTE immunotherapy with a longer half-life capable of being administered at longer dosing intervals. Preliminary results from the AMG 160 Phase I study showed that 34% of patients responded to PSA50 with adverse effects such as CRS. However, mitigation strategies used during the study could reduce the rate of adverse reactions.
>>Click here to explore more bispecific antibody target proteins and download the free protocols
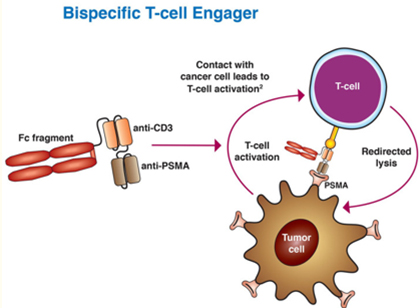
Image Source: PSMA targeting in metastatic castration-resistant prostate cancer: where are we and where are we going? Doi: 10.1177/17588359211053898
PSMA has attained attention recently as a tumor marker specific to the surface of prostate cell membranes, and bispecific antibody, RLT, and CAR-T therapies targeting PSMA have appeared. From the current research and development progress. Beyond vaccine therapies and checkpoint blockade, new therapies, such as ADCs, BiTEs, and CAR-T cells, offer intriguing and promising avenues for the treatment of advanced prostate cancer.
ACROBiosystems has been deeply involved in the field of recombinant proteins for many years and has developed a series of high-quality PSMA proteins to accelerate PSMA-related research.
![]() Various species:Human, Mouse, Cynomolgus, Rat;
Various species:Human, Mouse, Cynomolgus, Rat;
![]() Various tags:Fc, His, Fc & Avi;
Various tags:Fc, His, Fc & Avi;
![]() A wide range of application: Immune / antibody screening/cell based assay, etc.
A wide range of application: Immune / antibody screening/cell based assay, etc.
![]() High purity:SDS-PAGE verification purity>95%
High purity:SDS-PAGE verification purity>95%
SEC-MALS verification purity>90%;
![]() High bioactivity verified by ELISA/SPR/FACS. Free protocol offered;
High bioactivity verified by ELISA/SPR/FACS. Free protocol offered;
| Cat. No. | Species | Product Description | Preorder/Order |
|---|---|---|---|
| PSA-HP2Q3 | Human | PE-Labeled Human PSMA / FOLH1 Protein, His Tag (Site-specific conjugation) | |
| PSA-HF244 | FITC-Labeled Human PSMA / FOLH1 Protein, His Tag | ||
| PSA-H82Qb | Biotinylated Human PSMA / FOLH1 Protein, His,Avitag™ (active enzyme, MALS verified) | ||
| PSA-H52H3 | Human PSMA / FOLH1 Protein, His Tag (active enzyme, MALS verified) | ||
| PSA-H5264 | Human PSMA / FOLH1 Protein, Fc Tag | ||
| PSA-M5245 | Mouse PSMA / FOLH1 Protein, His Tag (active enzyme) | ||
| PSA-R5245 | Rat | Rat PSMA / FOLH1 Protein, His Tag (active enzyme, MALS verified) | |
| PSA-C5247 | Cynomolgus | Cynomolgus PSMA / FOLH1 Protein, His Tag (MALS verified) (active enzyme) |
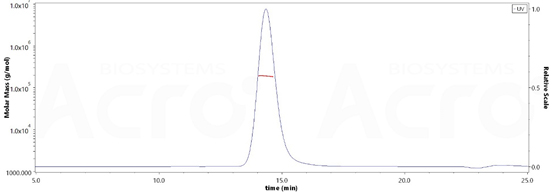
The purity of Rat PSMA, His Tag (Cat. No. PSA-R5245) is more than 90% and the molecular weight of this protein is around 170-208 kDa verified by SEC-MALS.
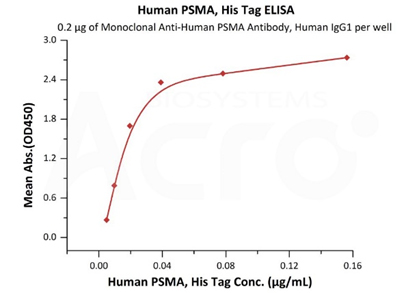
Immobilized Monoclonal Anti-Human PSMA Antibody, Human IgG1 at 2 μg/mL (100 μL/well) can bind Human PSMA, His Tag (Cat. No. PSA-H52H3) with a linear range of 2-39 ng/mL (QC tested).
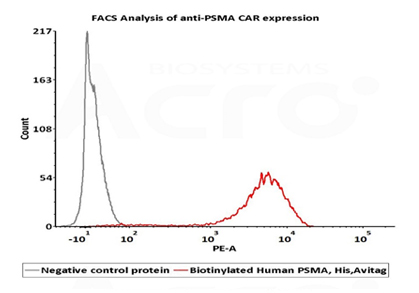
2e5 of PSMA-CAR-293 cells transfected with anti-PSMA-scFv were stained with 100μl of 1μg/mL of Biotinylated Human PSMA, His,Avitag (Cat. No. PSA-H82Qb) and negative control protein respectively, washed and then followed by PE-SA and analyzed with FACS (Routiney tested).
Fay, E. K., & Graff, J. N. (2020). Immunotherapy in Prostate Cancer. Cancers, 12(7), 1752. https://doi.org/10.3390/cancers12071752
Wang, F., Li, Z., Feng, X. et al. Advances in PSMA-targeted therapy for prostate cancer. Prostate Cancer Prostatic Dis (2021). https://doi.org/10.1038/s41391-021-00394-5
This web search service is supported by Google Inc.
