> Fc Receptor Proteins 
The efficacy of a therapeutic antibody not only depends on the Fab fragment and its binding activity to the target antigen, but also depends on the Fc fragment and its interaction with key Fc receptors. The binding affinity of the Fc fragment towards FcRn (FCGRT&B2M) would predict the antibody’s half-life, while that between the Fc fragment and FcγRIIIa (CD16a) would influence the antibody’s ability to elicit ADCC (antibody dependent cell mediated cytotoxicity). Therefore, candidates must be tested against a panel of receptors during antibody engineering.
ACROBiosystems offers a comprehensive collection of recombinant Fc receptor proteins, including their common variants, to expedite your antibody drug development.
![]() Expressed by HEK293 Cells: post-translational modification and proper protein folding
Expressed by HEK293 Cells: post-translational modification and proper protein folding
![]() Multiple species: Human, Mouse, Cynomolgus/Rhesus macaque, Rat, Porcine, Rabbit, Feline, Bovine, can be fully applied to different cross species experiments
Multiple species: Human, Mouse, Cynomolgus/Rhesus macaque, Rat, Porcine, Rabbit, Feline, Bovine, can be fully applied to different cross species experiments
![]() High purity: SDS-PAGE verification purity>95%, SEC-MALS verification purity>90%
High purity: SDS-PAGE verification purity>95%, SEC-MALS verification purity>90%
![]() Low endotoxin: <1.0 EU/µg
Low endotoxin: <1.0 EU/µg
![]() High stability: strict quality control to ensure high batch-to-batch consistency
High stability: strict quality control to ensure high batch-to-batch consistency
![]() Biotinylated Fc Receptor proteins labeled with AvitagTM offered: the labeling efficiency is high, and the labeling site is specific and clear, which is suitable for ELISA/SPR/BLI detection based on binding to streptavidin in the process of drug development and optimization process
Biotinylated Fc Receptor proteins labeled with AvitagTM offered: the labeling efficiency is high, and the labeling site is specific and clear, which is suitable for ELISA/SPR/BLI detection based on binding to streptavidin in the process of drug development and optimization process
![]() Affinity verified by SPR & BLI: activity guaranteed, and protocols offered free of charge
Affinity verified by SPR & BLI: activity guaranteed, and protocols offered free of charge
To meet the high purity requirement of pharmaceutical applications, our production team perform both SDS-PAGE and SEC-MALS analyses to the Fc receptors for QC purposes. It’s noteworthy that it’s essential to use monomeric Fc receptor, which represents its natural state. The formation of oligomers during production may result in artificially enhanced binding interaction due to avidity effect. To avoid such situation, we have established strict internal control standards of monomer purity by SEC-MALS testing. Only those batches meeting all purity requirements are released.
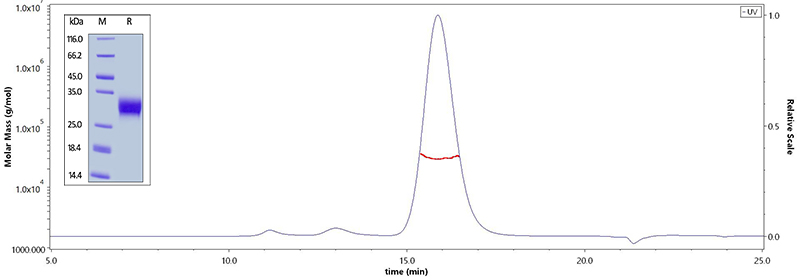
Fig. 1 The purity of Human CD32a (R167), His Tag (Cat. No. CDA-H5221) is more than 95% verified by SDS-PAGE. The purity of Human CD32a (R167), His Tag (Cat. No. CDA-H5221) is more than 90% verified by SEC-MALS. And the molecular weight of this protein is around 25-35 kDa.
The binding affinity between two molecules can be determined by many methods. However, the studies of Fc interaction are often done by SPR, because traditional ELISA is not appropriate for low affinity receptors such as CD16 and CD32. To assure our Fc receptors deliver expected performance, our QC team use Biacore platform to test our products against reference antibodies. Additionally, to meet more applications in the development of antibody drugs, our QC team also use ForteBio platform to test our products against reference antibodies. All SPR&BLI protocols are available for free.
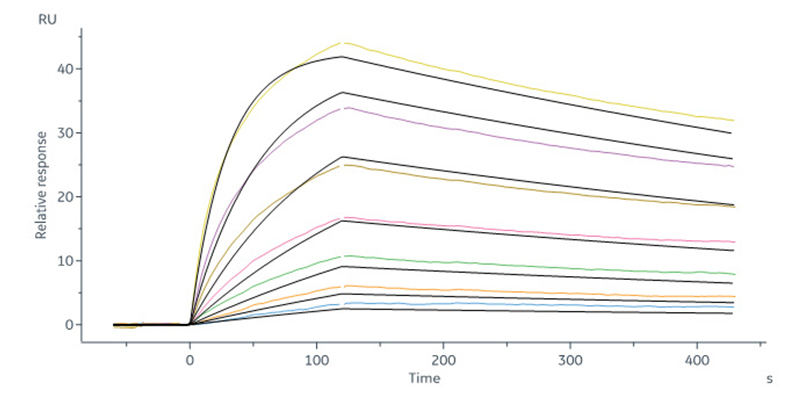
Fig. 2 Human CD64, His Tag (SPR & BLI verified) (Cat. No. FCA-H52H1) captured on CM5 chip via anti-His antibody can bind Herc*ptin with an affinity constant of 4.92 nM as determined in a SPR assay (Biacore 8K) (QC tested).
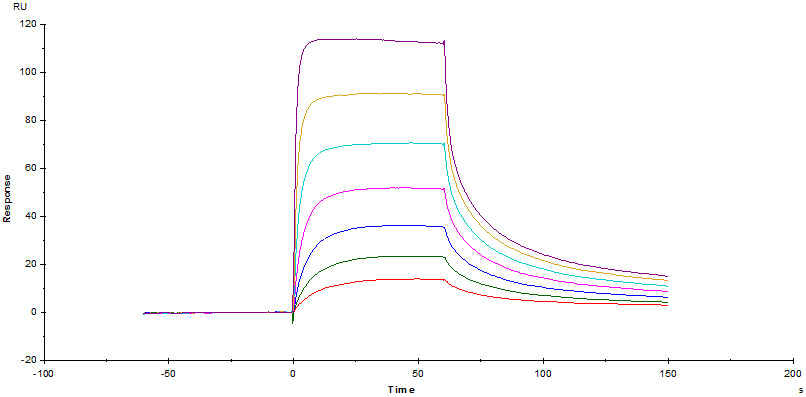
Fig. 3 Immobilized Human FCGRT&B2M Heterodimer Protein, His Tag&Strep II Tag (SPR & BLI verified) (Cat. No. FCM-H5286) on CM5 Chip via Anti-His antibody, can bind Herc*ptin with an affinity constant of 0.261 μM as determined in a SPR assay (Biacore T200).

Fig. 4 Loaded Human CD64, His Tag (SPR & BLI verified) (Cat. No. FCA-H52H1) on HIS1K Biosensor, can bind Herc*ptin with an affinity constant of 6.98 nM as determined in BLI assay (ForteBio Octet Red96e).
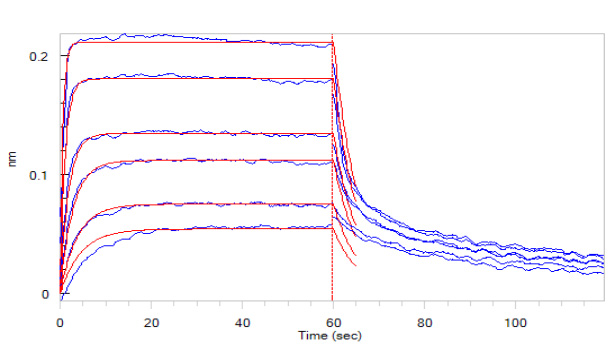
Fig. 5 Loaded Human FCGRT&B2M Heterodimer Protein, His Tag (SPR & BLI verified) (Cat. No. FCN-H52W7) on SA Biosensor via Biotin his antibody, can bind Herc*ptin with an affinity constant of 0.14 μM as determined in BLI assay (ForteBio Octet Red96e).
We routinely apply rigorous quality control measures to ensure consistent performance of our product. As shown below, the batch variation among the tested samples is negligible.
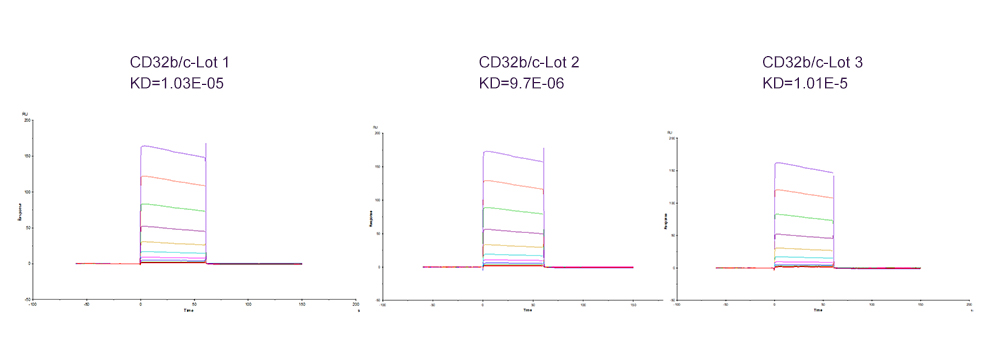
Fig. 6 Immobilized Human Fc gamma RIIB / CD32b Protein (Cat. No. CDB-H5228) on CM5 Chip via anti-His antibody, can bind Ritux*mab with an affinity constant of 10 μM as determined in a SPR assay (Biacore T200).
We have compared the performance of different batches of CD32b (CDB-H5228) in the same assay, and the software analysis showed that the similarity score was very high, meaning high batch-to-batch consistency.
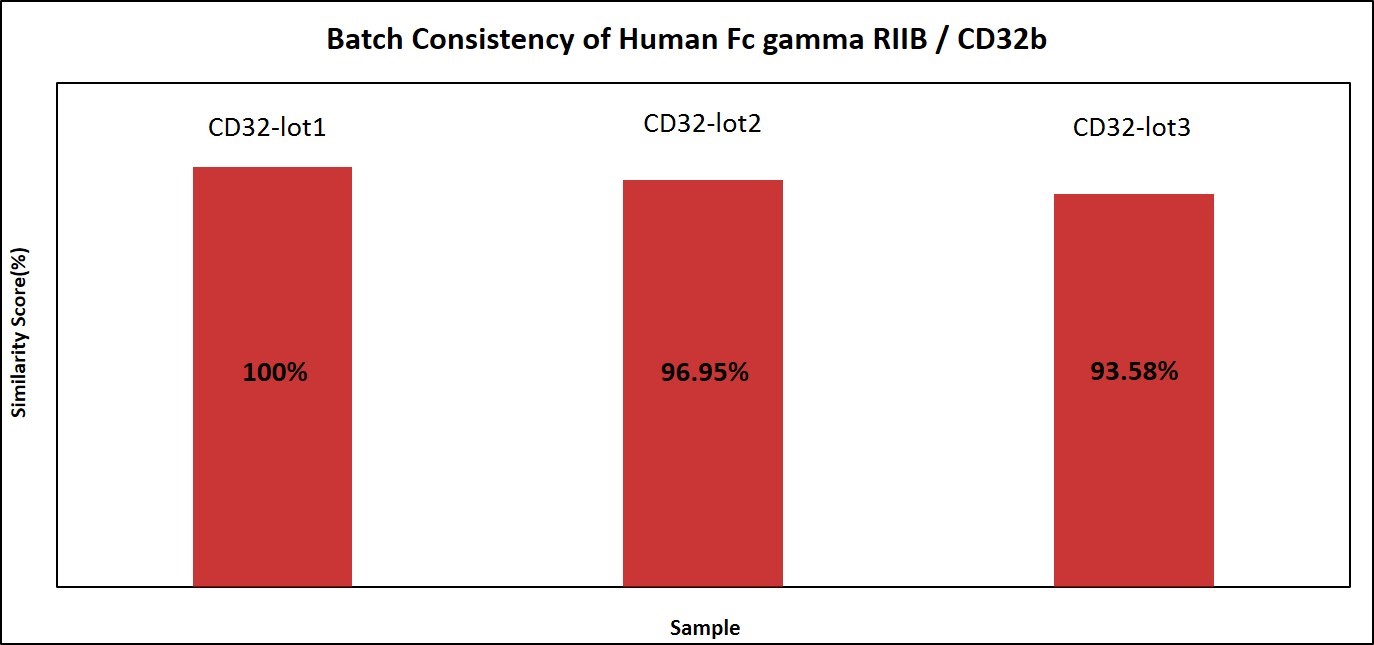
Fig. 7 Batch consistency of Human Fc gamma RIIB / CD32b (Cat. No. CDB-H5228). The consistency of different batches is more than 90%.
The use of biotin labeling can make your assay development much easier. We offer a variety of ready-to-use biotinylated Fc receptors. These proteins are produced using our in-house developed labeling techniques, which confers high bioactivity and minimal batch-to-batch variation. In addition, we have developed a series of Fc receptors of other species, which are suitable for the screening of non-humanized antibodies or the species cross reaction.
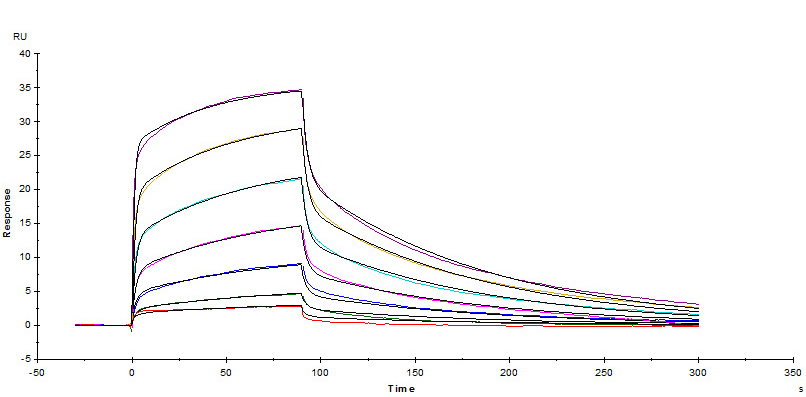
Fig. 8 Biotinylated Human CD16a (V176), His Tag, Avi Tag (Cat. No.CDA-H82E9) captured on Biotin CAP- Series S Sensor Chip can bind Ritux*mab with an affinity constant of 0.261 μM as determined in a SPR assay (Biacore T200).
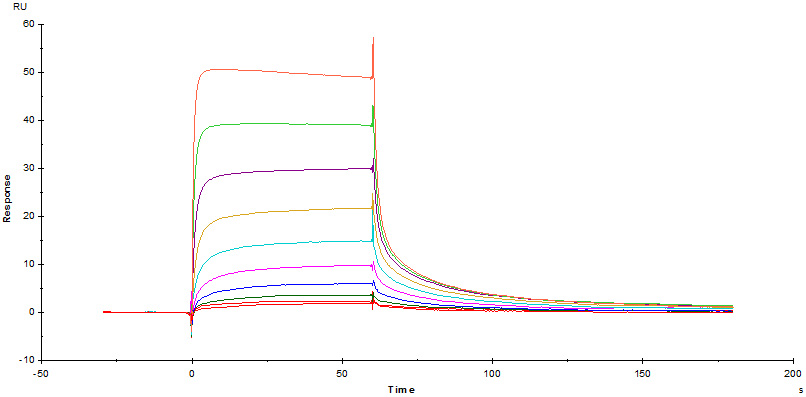
Fig. 9 Immobilized Cynomolgus / Rhesus macaque FcRn Protein (Cat. No. FCM-C5284) on CM5 Chip via anti-His antibody, can bind Herc*ptin with an affinity constant of 0.403 μM as determined in a SPR assay (Biacore T200).
FcRn
FcγR
FcεR
| Molecule | Cat. No. | Host | Product Description | Structure |
|---|
| Molecule | Cat. No. | Host | Product Description | Structure |
|---|
| Molecule | Cat. No. | Host | Product Description | Structure |
|---|
Authors: Kielau Kisalu, Neville et al.
Journal: JCI Insight 2020
Cited Product: FCM-H5286,FCM-C5284,FCM-M82W5
Application: BLI
Authors: Asokan M, Dias J, Liu C, et al
Cited Product: FCM-C5284
Journal: Proc Natl Acad Sci U S A2020
Application: BLI
Authors: Simonov V, et al
Cited Product: FCM-H5286
Journal: Biologicals 2019
Application: BLI
Engineered antibody Fc variants for enhanced serum half life
Authors: G Georgiou, et al
Cited Product: FCM-H5286
Journal: US20190048078A1 2019
Application: SPR
Authors: Yang B, et al.
Cited Product: FCM-H5286,FCM-M52W2
Journal: Biomater Sci 2018
Application: ELISA
Authors: Zhao C, et al.
Cited Product: FCM-H5286
Journal: Mol Cell Endocrinol 2018
Application: SPR
Authors: Kenniston JA, et al.
Cited Product: FCM-H5286,FCM-M52W2,FCM-R5287,FCM-C5284
Journal: J Biol Chem 2017
Application: ELISA & SPR
Single chain Fc-dimer-human growth hormone fusion protein for improved drug delivery
Authors: Li Zhou, et al.
Cited Product: FCM-H5286
Journal: Biomaterials 2016
Application: SPR
CD38 ecto-enzyme in immune cells is induced during aging and regulates NAD + and N*N levels
Authors: Chini CCS, Peclat TR, Warner GM, et al.
Cited Product: FC4-M52H3,CD4-M5227
Journal: Nature metabolism 2020
Application: Binding Assay
Authors: Simonov V, et al
Cited Product: CDB-H5228,CDA-H5221,CDA-H5220,FCA-H52H2,CDB-H5222
Journal: Biologicals 2019
Application: SPR
Identification of anti-CD16a single domain antibodies and their application in bispecific antibodies
Authors: Zhao Y, et al
Cited Product: CDA-H5220
Journal: Cancer Biol Ther 2019
Application: immunization
Charge variant analysis of proposed biosimilar to Trastuz*mab
Authors: Dakshinamurthy P, et al.
Cited Product: CD8-H52H4
Journal: Biologicals 2017
Application: SPR
Sialylation of immunoglobulin E is a determinant of allergic pathogenicity
Authors: Kai Ting C Shade, et al
Cited Product: CD3-H5249
Journal: Nature 2020
Application: BLI
> Learn more about ComboX, a combination of universal solutions
This web search service is supported by Google Inc.
