> Biotinylated Protein 
The interaction between biotin and avidin/streptavidin (SA) is incredibly strong and specific, making it an excellent tool in protein biochemistry. However, generation of high quality biotinylated proteins presents a major challenge. Researchers often run into technical issues including insufficient labeling, high batch-to-batch variation, and unpredictable performance. In addition, the choice of biotinylation methods also significantly affects the ultimate outcome. Each target protein (assay) has its own unique biochemical characteristics, and therefore requires customized solution.
These confounding factors make it difficult for individual labs to make and use biotin conjugates, while at the same time few products are available commercially. To fill in this gap, ACROBiosystems has developed an exclusive collection of pre-labeled and experimentally validated biotinylated recombinant proteins. The Mabsol® biotinylated protein collection includes more than a hundred commonly studied drug targets and biomarker proteins.
We use high quality recombinant proteins that are closest to natural conformation and modification as starting materials. We thoroughly go through every option to achieve the highest bioactivity and detection sensitivity. We also apply the most stringent quality control measurement possible to ensure minimal batch-to-batch variations.
Mabsol® biotinylated protein collection includes two unique and complimentary product series, the PrecisionAvi series built upon the AvitagTM technology (See full list) , and the UltraLys series produced using the in-house developed chemical labeling method (See full list). These products are made with every attention to details.
The products in this series are exclusively produced using the AvitagTM technology. Briefly, a unique 15 amino acid peptide, the Avi tag, is introduced into the recombinant protein during expression vector construction. The single lysine residue in the Avi tag is enzymatically biotinylated by the E. Coli biotin ligase BirA.
This single-point enzymatic labeling technique brings many advantages for commonly used binding assays.
The biotinylation only happens on the lysine residue of Avi tag.
NO interference with the target protein’s natural binding activities.
The protein orientation is uniform when immobilized on an avidin-coated surface.
The products in this series are produced using our in-house developed chemical labeling approach. The primary amines in the side chains of lysine residues and the N-terminus of protein are conjugated with biotins, usually resulting in multiple biotin attachment on a single protein molecule.
Higher detection sensitivity
In-house developed chemical labeling approach
The production of our recombinant proteins including the biotinylated proteins is carried out using our proprietary HEKMax® expression platform. As expression hosts, the human HEK293 cells have a variety of advantages compared to other cell types as summarized in the table below.
The bioactivity of biotinylated proteins is determined both by the structure of the protein itself, and by the way how biotinylation is performed. For every single protein, we test multiple options of tags and biotinylation methods, and evaluate the products in a variety of binding assays. Only those with the best performance are selected for production. Figure 1 is an example of our internal evaluation experiments:
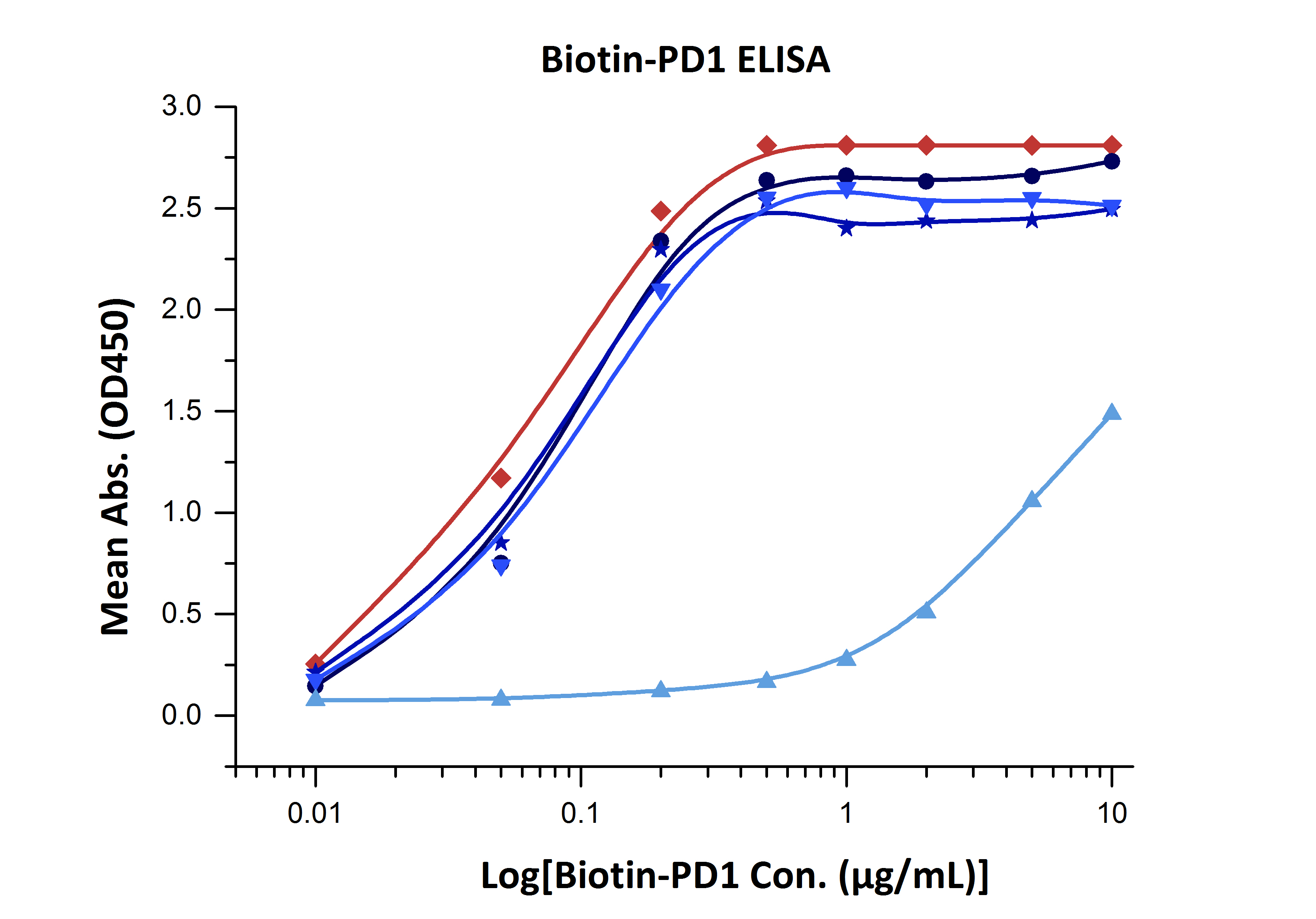
Fig. 1 Binding activity of different forms of biotinylated PD1 evaluated in a functional ELISA against rhPD-L1 (Cat. No. PD1-H5258).
Our biotinylated proteins demonstrate high bioactivity and superb detection sensitivity in different application. In functional ELISA, the biotinylated human TACI has high binding activity with BAFF as showed in Fig. 2. Interestingly, oriented immobilization of biotinylated CD155 offers much higher sensitivity than random immobilization of unconjugated CD155 as showed in Fig. 3. Fig.4 and Fig. 5 show that the biotinylated PD1 can be well applied to AlphaLISA to detect the binding activity with its ligand PD-L1 and PD-L2. The Fig. 6 and Fig. 7 show that the biotinylated protein can be easily used for determining the affinity between protein and therapeutic antibody in SPR assay. Also, the biotinylated proteins are often used in cell based assay, such as cytotoxicity assay and evaluation of CAR expression (Fig. 8 & Fig. 9).

Fig.2 Immobilized Human BAFF, Fc Tag, active trimer (Cat. No. BAF-H5261) at 1 μg/mL (100 μL/well) can bind Biotinylated Human TACI, Fc,Avitag (Cat. No. TAI-H82F6) with a linear range of 0.002-0.05 μg/mL.
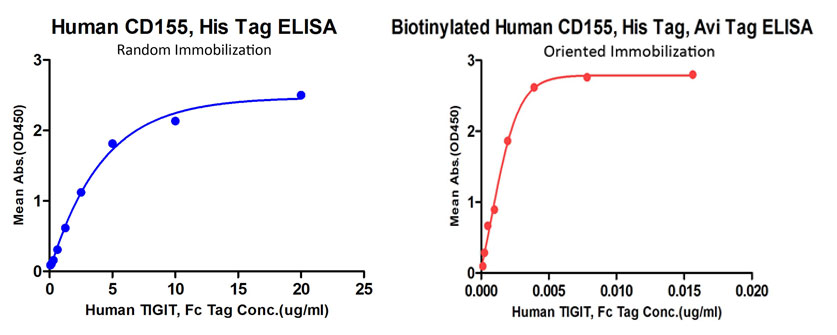
Fig. 3 Comparison of oriented immobilization (biotinylated CD155 on precoated streptavidin plate, Cat. No. CD5-H82E3) and random immobilization (unconjugated CD155 direct coating on 96-well plate, Cat. No. CD5-H5223)binding to TIGIT( Cat. No. TIT-H5254). Oriented immobilization of biotinylated CD155 offers much higher sensitivity than random immobilization of unconjugated CD155.

Fig. 6 Immobilized biotinylated human VEGF165 (Cat. No. VE5-H82Q0) on CM5 Chip via streptavidin, can bind Avastin with an affinity constant of 0.417nM as determined in SPR assay (Biacore T200).

Fig.7 Loaded Biotinylated Human PD-1, Avitag,His Tag (recommended for biopanning) (Cat. No. PD1-H82E4) on SA Biosensor, can bind Human PD-L1, His Tag (HPLC verified) (Cat. No. PD1-H5229) with an affinity constant of 2.4 μM as determined in BLI assay (ForteBio Octet Red96e).
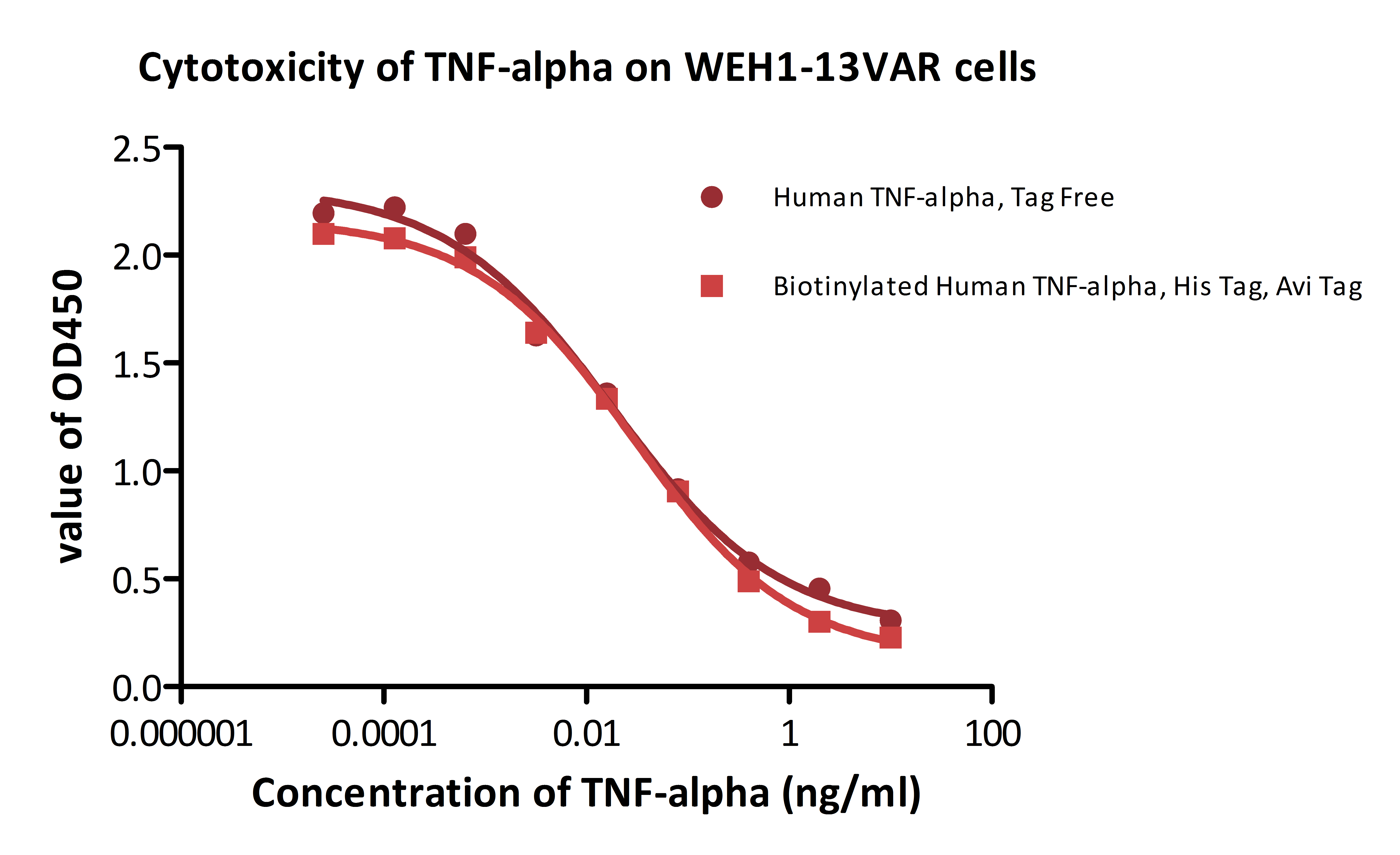
Fig. 8 Recombinant human TNF-alpha (Cat. No. TNA-H82E3) induces cytotoxicity effect on the WEH1-13VAR cells in the presence of the metabolic inhibitor actinomycin D. The EC50 for this effect is 0.014-0.029 ng/ml. The result shows that the biotinylated human TNF-alpha is consistent with naked TNF-alpha in cytotoxicity assay.
We routinely apply rigorous quality control measures to ensure consistent performance of our products. Newly produced products are subjected to side-by-side comparison with our internal standard in a variety of assays. Only those within an acceptable margin of difference are allowed to be released. As demonstrated in Fig. 9, three different batches of biotinylated hTNF-alpha (Cat. No. TNA-H82E3) are tested and compared using a standard ELISA analysis against Adalim*mab, the result shows that the batch variation among the tested samples is negligible. The batch-to-batch conformance is also good as displayed in cell based assay (Fig. 10).
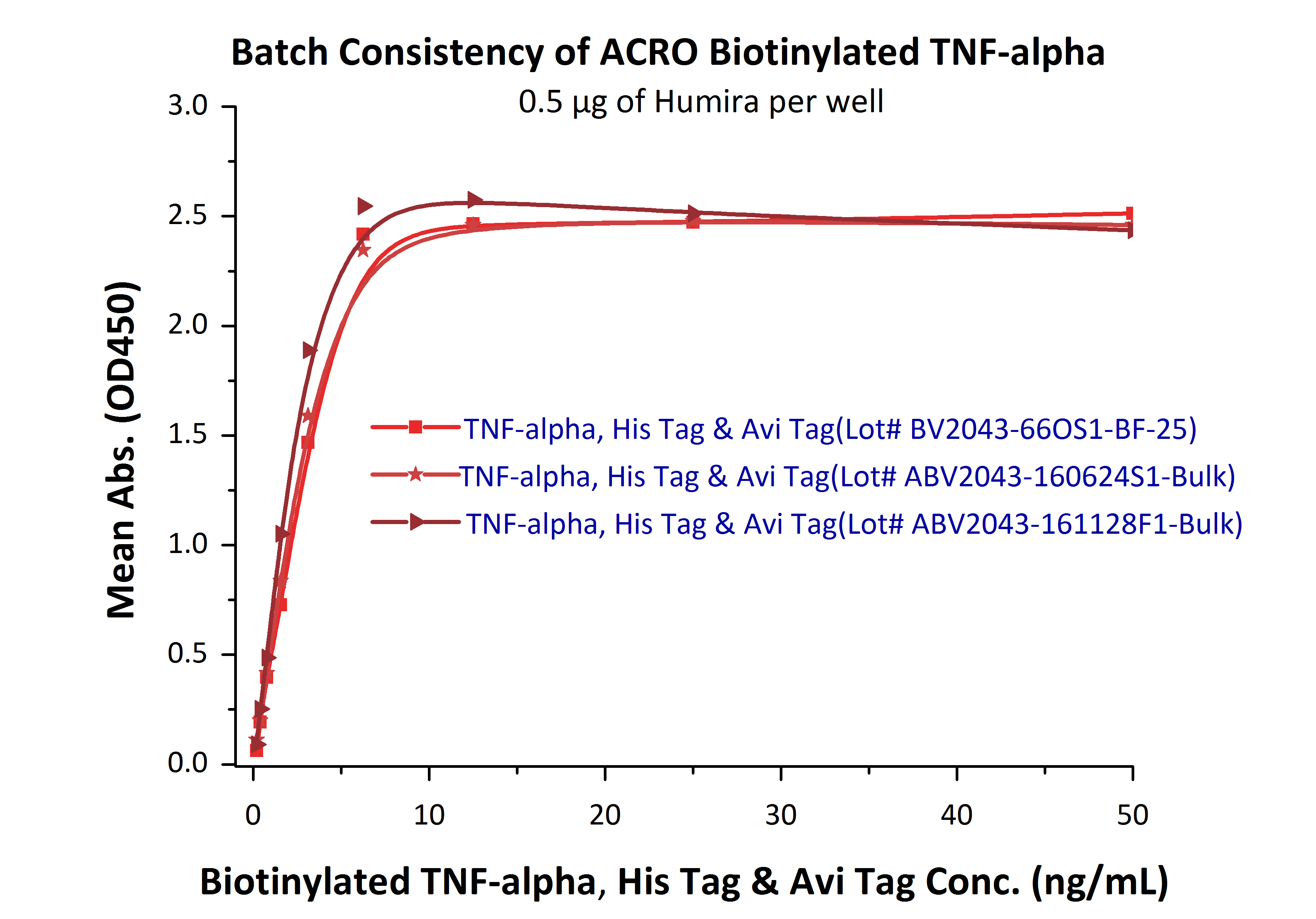
Fig. 10 In the above ELISA analysis, three different lots of biotinylated hTNF-alpha (Cat. No. TNA-H82E3) were used detect immobilized Adalim*mab (0.5 ug/ml). The result showed that the batch variation among the tested samples is negligible.
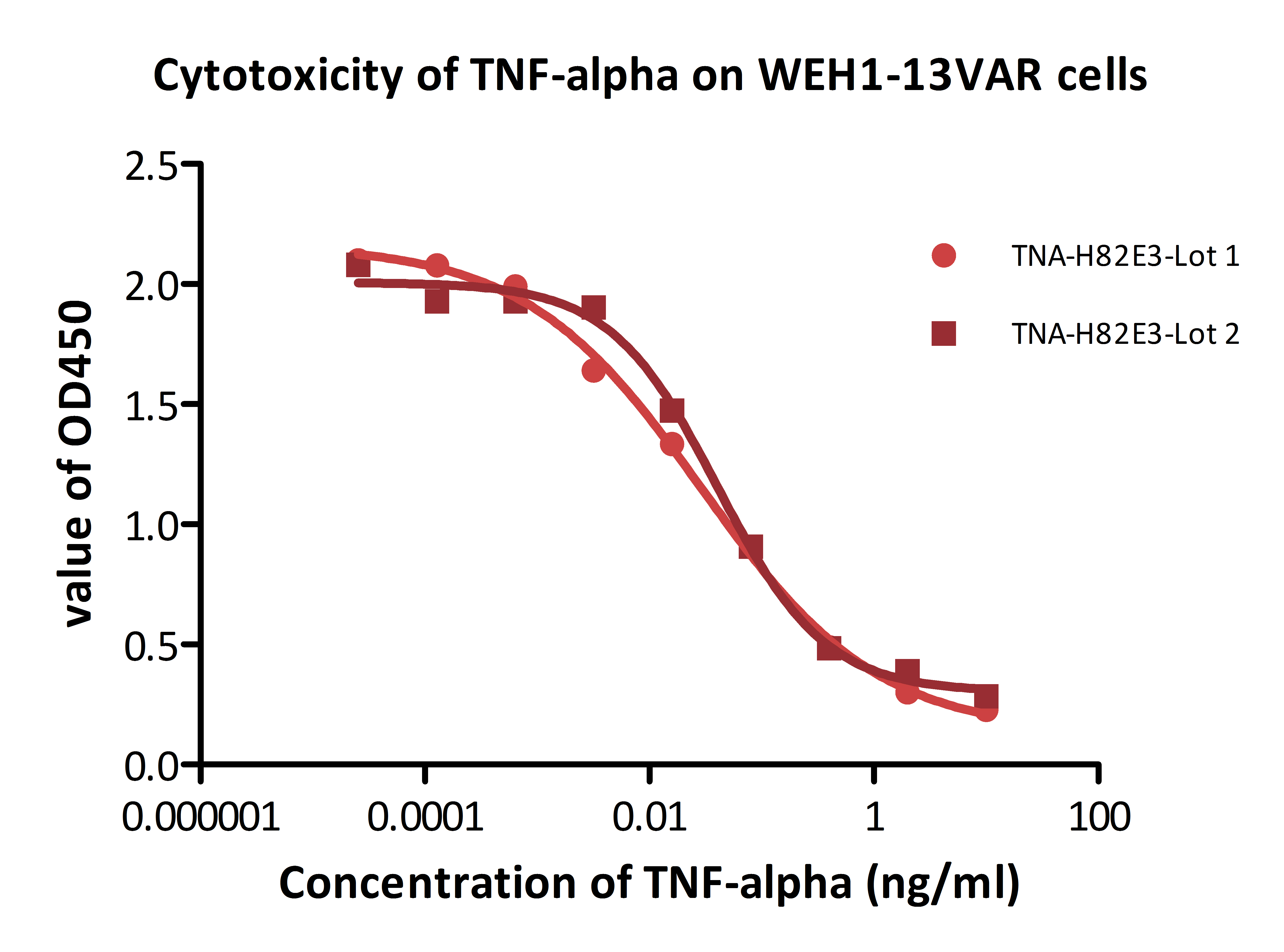
Fig. 11 Recombinant biotinylated human TNF-alpha (Cat. No. TNA-H82E3) induces cytotoxicity effect on the WEH1-13VAR cells in the presence of the metabolic inhibitor actinomycin D. The EC50 for this effect is 0.029-0.052 ng/ml.
Authors: MacLeod DT et al.
Journal: Mol Ther 2017
Product: biotinylated Human CD19(Cat. No. CD9-H8259)
Application: Flow Cytometry
Authors: El Amrani M et al.
Journal: J Chromatogr A 2016
Product: biotinylated human recombinant TNF-alpha(Cat. No. TNA-H8211)
Application: ELISA
Authors: Kim Young Min et al.
Journal: United States Patent Application 20160136300 2016
Product: ErbB2(Cat. No. HE2-H822R)
Application: ELISA
Authors: Grzeschik J et al.
Journal: Biotechnol J 2016
Product: EpCAM(Cat. No. EPM-H8223)
Application: Flow Cytometry
Authors: Pooja Sabhachandani et al.
Journal: Microchimica Acta 2015
Product: biotinylated human TNF-α(Cat. No. TNA-H8211)
Application: Microsphere-based assay
Authors: Zielonka S et al.
Journal: Marine Biotechnology 2015
Product: CD3 epsilon(Cat. No. CDE-H8223)
Application: FACS sorting
Authors: YU et al.
Journal: United States Patent Application 20140155285 2014
Product: biotinylated TNFα(Cat. No. TNA-H8211)
Application: Flow Cytometry
This web search service is supported by Google Inc.
